Metals and advances in manufacturing processes gave us the Industrial Revolution. This led to the exponential growth of human civilisation, bringing us where we are today. Today, different types of metals are all around us. From the computer you are using to read this information to the clamps in your plumbing. More than eighty different types of metals find use today.
Types of Metal and Their Classification
A large number of metals are available in nature. They can be classified in a variety of ways depending on what property or characteristic you use as a yardstick.
Classification by Iron Content
The most common way of classifying them is by their iron content.
When a metal contains iron, it is known as a ferrous metal. The iron imparts magnetic properties to the material and also makes them prone to corrosion. Metals that do not have any iron content are non-ferrous metals. These metals do not possess any magnetic properties. Examples include but are not limited to aluminium, lead, brass, copper and zinc.
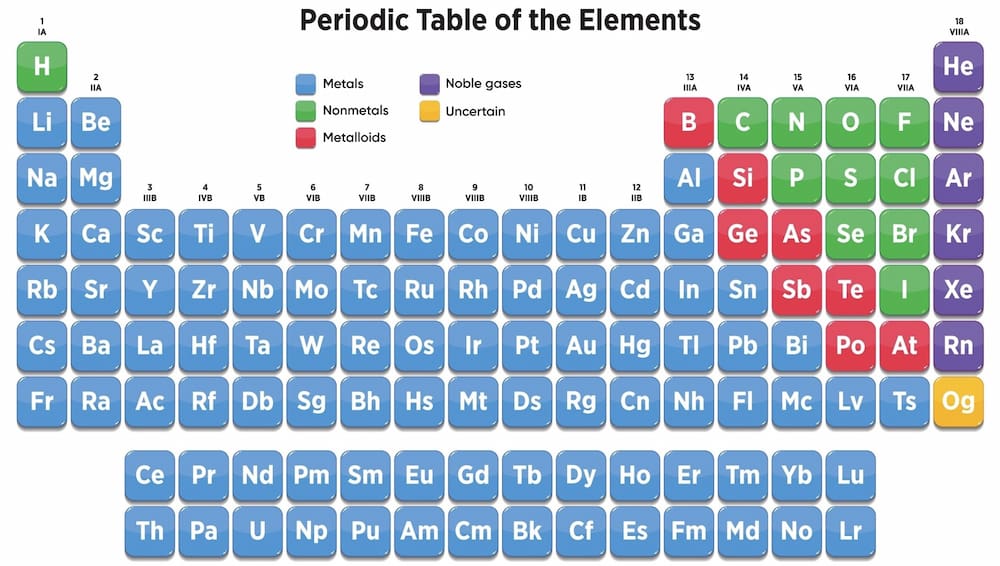
Classification by Atomic Structure
They may also be classified based on their atomic structure according to the periodic table. When done, a metal may be known as an alkaline, alkaline earth, or a transition metal. Metals belonging to the same group behave similarly when reacting with other elements. Thus, they have similar chemical properties.
Magnetic and Non-Magnetic Metals
Another way to differentiate metals is by looking at how they interact with magnets. It is possible to divide metals into magnetic and non-magnetic on that basis.
While ferromagnetic metals attract strongly to magnets, paramagnetic ones only show weak interactions. Lastly, there is a group called diamagnetic metals that show a rather weak repulsion to magnets.
Iron, Its Alloys and Their Properties
All the metals share some similar mechanical properties of materials. But when judged closely, one metal will have a slight edge over another in certain properties. It is possible to tweak the properties when creating alloys by mixing pure elements.
When selecting a metal for a particular application, there are quite a few factors to consider to find the most suitable option. These factors include melting point, cost, ease of machining, sufficient safety factor, space available, temperature coefficient, thermal and electrical conductivity, density, etc. Let us take a look at some of the popular metals and why they are chosen for their applications.
Iron

It would not be an exaggeration to refer to iron as the lifeblood of our civilisation. Approximately 5 per cent of the Earth’s crust is iron. Thus, it is an incredibly easy metal to find. Pure iron is an unstable element, though. At the first opportunity, it reacts with the oxygen in the air to form iron oxide.
Extracting iron from its ores uses a blast furnace. Pig iron is obtained from the first stage of the blast furnace, which can be further refined to obtain pure iron. This iron often ends up in steels and other alloys. Almost 90 per cent of manufactured metals are ferrous metals.
Steel, for instance, is a ferrous metal that finds a variety of applications. We cannot comprehend the true potential of iron without learning about steel.
Steel
Pure iron is stronger than other metals, but it leaves much to be desired. For one, pure iron is not resistant to corrosion. To keep iron from corroding, a lot of money and energy must be spent. Secondly, it is also extremely heavy due to its high density. These disadvantages can make structures harder to build and maintain.
Adding carbon to iron alleviates these weaknesses to a certain extent. This mixture of iron and carbon up to specified limits is known as carbon steel. Adding carbon to iron makes the iron much stronger, along with imparting other great characteristics.
Other elements may be added in trace amounts to incorporate their properties. Let’s take a look at how to categorise steel and what it is capable of.
- Personal account manager
- Quality assurance
- Payment terms for companies
- On-time delivery by Fractory
What are the Types of Steel and Their Uses?
Steel is a popular building material thanks to its excellent properties. Over 3500 grades of steel are available today. It has high tensile strength and a high strength-to-weight ratio. This means more strength per unit mass of steel. This allows the usage of steel parts and components that are small in size but still strong.
Steel is also extremely durable. This means a steel structure can last longer and withstand external factors better than other alternatives. It is also ductile and can be shaped into required forms without compromising its properties. Depending on the iron content, steel is classified into three categories.
Carbon Steel AISI Classification

- Low carbon steel. Up to 0.25% of carbon in iron gives us low carbon steel, also known as mild steel. It is used for tubing in moderate-pressure applications. Reinforcing bars and I-beams in construction are usually from low carbon steel. Any applications that require a high amount of steel without much forming or bending are also suitable for it. An example is a ship’s hull.
- Medium carbon steel. Contains 0.25…0.6% of carbon. Medium carbon steel’s applications include ones that need high tensile strength and ductility. They find applications in gearing and shafts, railway wheels and rails, steel beams in buildings and bridges, etc. Another use is pressure vessels, except if they contain cold gases or liquids, because of its tendency to cold cracking.
- High carbon steel. Steel that contains more than 0.6% carbon is high carbon steel. This steel is harder and more brittle than the previous two. It finds applications in making chisels and cutting tools. Great qualities include hardness and good resistance to material wear. It may also be used in presses and for manufacturing drill bits.
Although all the above-mentioned steels are commonly referred to as carbon steels, they contain other elements to improve certain properties. Like chromium for corrosion resistance or manganese to improve hardenability and tensile strength.
Alloy steels
This type of metal contains multiple elements to enhance various properties. Metals such as manganese, titanium, copper, nickel, and aluminium are common alloying agents—alongside silicon, a metalloid that enhances strength and magnetic properties.
In addition, alloying elements improve steel’s hardenability, weldability, corrosion resistance, ductility, and formability. Applications for alloy steels are electric motors, bearings, heating elements, springs, gears, and pipelines.
- Stainless steel: Stainless steel contains high amounts of chromium. This is why it has 200 times higher resistance to corrosion than mild steel. It makes it the ideal candidate to manufacture kitchen utensils, piping, surgical, and dental equipment. Also, as no coating is necessary, you can have a metallic look like you want with the right surface finish.
- Tool steel: Tool steels are used for making cutting and drilling tools. Their high hardness makes them an ideal choice for these applications. They contain molybdenum, vanadium, cobalt, and tungsten as constituent metals.

Tool steel is a type of metal that also finds applications in manufacturing rails, wires, pipes, shafts, and valves. Tool steel is primarily used in the automotive, shipbuilding, construction, and packaging sectors.
Different Types of Metals
In addition to ferrous metals, we have a large selection of non-ferrous ones. Each has certain qualities that make them useful in different industries.
Aluminium
Aluminium derives primarily from its ore, bauxite. It is light, strong and functional. It is the most widespread metal on Earth and its use has permeated applications everywhere.
This is because of its properties such as durability, lightweight, corrosion resistance (learn more about the types of aluminium corrosion here), electrical conductivity and ability to form alloys with most metals. It also doesn’t magnetise and is easy to machine.
Copper
When talking about different types of metals, copper and its alloys can not be overlooked. It has a long history because it is easy to form. Even today, it is an important metal in the industry. It does not occur in nature in its pure form. Thus, smelting and extracting from ore is necessary.
Metals are good conductors, and copper stands out more than the others. Due to its excellent electrical conductivity, it finds application in electrical circuits as a conductor. Its conductivity is second only to silver. It also has excellent heat conductivity. This is why many cooking utensils are from copper.
Copper exhibits a unique form of corrosion that leads to the formation of a distinctive blue-green patina, which not only affects its aesthetic appeal but also serves as a protective layer against further corrosion.
Brass

Brass is an alloy of copper and zinc. The amount of each of the metals may vary depending on the electrical and mechanical properties sought of the metal. It also contains trace amounts of other metallic elements such as aluminium, lead, and manganese. Brass is a great candidate for low friction applications such as locks, bearings, plumbing, musical instruments, tools and fittings. It is indispensable in intrinsically safe applications to prevent sparks and allow usage in flammable environments.
Bronze
Bronze is also an alloy of copper. But instead of zinc, bronze contains tin. Adding other elements such as phosphorus, manganese, silicon, and aluminium may improve its properties and suitability for a particular application. Bronze is brittle, hard, and resists fatigue well. It also has good electrical and thermal conductivity and corrosion resistance. Bronze finds application in the manufacturing of mirrors and reflectors. It is used for electrical connectors. Due to its corrosion resistance, it finds usage in submerged parts and ship fittings.
Titanium
Titanium is an important engineering metal due to being strong and lightweight. It also has high thermal stability even at temperatures as high as 480 degrees Celsius. Due to these properties, it finds application in the aerospace industry. Military equipment is one use case for this metal. Since titanium is also corrosion-resistant, medical applications also use it. Titanium is also used in the chemical and sporting goods industry.
Zinc

Zinc is a widespread metal and finds a lot of use in the medical and industrial sectors. Its primary use is to galvanise steel. This protects the steel from corrosion. Zinc is also used to manufacture die castings for the electrical, hardware, and automobile industries. Since zinc has a low electrochemical potential, its uses include marine applications to prevent corrosion of other metals through cathodic protection. Sacrificial zinc anodes may protect valves, pipelines, and tanks.
Lead
Lead is a highly machinable, corrosion-resistant metal. Piping and painting represent some use cases. Lead was used as an anti-knocking agent in gasoline. Later, it was discovered that the byproduct of this lead was responsible for serious health complications. Lead is still common in ammunition, car batteries, radiation protection, lifting weights, cable sheathing, etc.
Fractory offers metal fabrication services, such as laser cutting, sheet metal bending and CNC machining, for a wide range of metals. If you don’t find suitable material on our platform, please contact our sales engineers, since they are most likely still able to accommodate your needs.



